The Catch 22 of R22 Replacements
FREON AND BEYOND
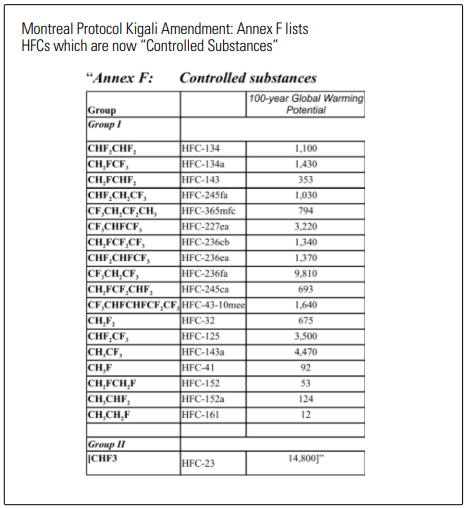
If you’ve recently googled “it’s almost 2020 and I still have R22” then hopefully this article grants you some timely insights. The world of refrigerants has become much more complex since you could refer to your refrigerant as “Freon” and not make an embarrassing social blunder because you were actually using a halogenated hydrocarbon and not a fluorinated hydrocarbon mixed with an unsaturated fluorinated alkene. While Freon® is simply a brand name which covers several legacy refrigerants (including R22) it continues to be the “kleenex” of the synthetic category despite there being many more brands to choose from which are wildly different in terms of application and performance.Things get more complex if you consider that the regulatory landscape has many R22 users retrofitting refrigerants before their system expires. This means compatibility of the new refrigerant with the old system is critical. What’s more is that while accounting for differences in capacity, efficiency, oil compatibility, operating pressures and temperatures, amongst several other factors, one must also attempt to predict the lifespan of the new gas. As R22, or more specifically, HCFCs (hydrochlorofluorocarbons), are phased out to protect the ozone, the HFC (hydrofluorocarbon) refrigerants available to replace them are now considered “super pollutants” contributing to the greenhouse effect. In 2016, the same mechanism used to phase out chlorine-containing refrigerants (eg R22) was revised to also regulate HFCs due to their global warming potential (GWP). The revision to the Montreal Protocol is the Kigali Amendment, which came into force on January 1, 2019. Although not all have chosen to ratify it yet, the signatories of the Kigali Amendment include 197 parties–which makes it truly a global resolution (there are only 195 countries in the world). In the United States, due to the current administration’s delay of Kigali ratification, more than half of the country (24 governors representing 55 percent of the US population and an $11.7 trillion economy) has joined what is called the US Climate Alliance which pledges to uphold the Paris Climate Agreement. This means agreement to reduce GHG (greenhouse gas) emissions–partly by passing legislation to phase down HFC refrigerants like California and Washington have already done, and several other states are in the process of doing.
As January 1, 2020, the conclusion date of the US R22 phase out, is now only months away, many are hustling to understand their next move. Those who have already replaced their ozonedepleting R22 with an HFC are now anxiously watching the patchwork of GHG legislation develop, while many who delayed their R22 conversions actually still have the luxury of considering ways to leap-frog HFCs to a more permanent or “future-proof” solution. It is therefore important to understand what refrigerant options are available and how they may perform based on their most fundamental characteristics.
REFRIGERANT PROPERTIES
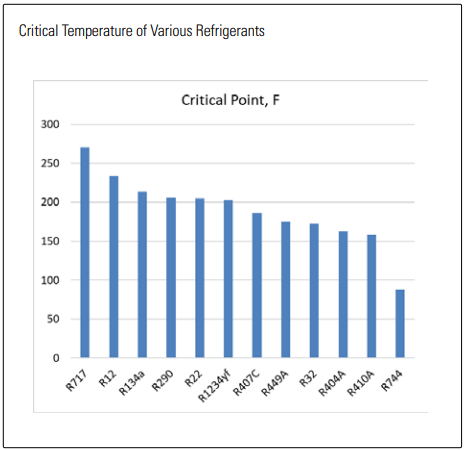
High critical temperature, high latent heat, low pressure ratio, and high refrigerating capacity per compressor displacement make for a good refrigerant that in turn make the refrigeration system both efficient and cost effective; however, not a single refrigerant wins in every category. Ammonia (R717) comes the closest as it has the best critical temperature and latent heat while having good capacity and low pressure ratio–which points to the reason ammonia has enjoyed great success over the past century and is still growing into new markets today. However, not every application is suited for ammonia, which is why R22 had been used primarily where ammonia had not.
CRITICAL TEMPERATURE OF VARIOUS REFRIGERANTS
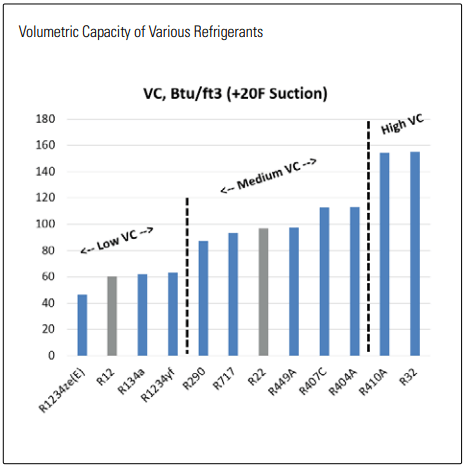
Today there are still a large number of R22 systems, many of which are nearing their end of life. Even the newer systems cannot revert to ammonia, but neither could they easily switch to carbon dioxide (CO2 ), propane, and probably most of the synthetics on the market. The latter is primarily due to one of the most important characteristics of a refrigerant: how much cooling it can produce given a certain amount of compressor displacement–referred to here as volumetric capacity (VC). For the purposes of this article, a “high” VC is with respect to the refrigerant, leading to a “low” compressor displacement requirement—and is a good thing. Broadly, refrigerants can be categorized into low, medium, and high VC groups, determined primarily by the latent heat and vapor density. Ammonia has a latent heat approximately 6 times that of other refrigerants, but a low vapor density counteracts much of this and yields a VC similar to that of R22. With CO2 the opposite is true in that an extremely high vapor density paired with an already good enthalpy gives it more than three times the VC of R32. This means CO2 compressors will be relatively small, however, they will need to overcome more than four times the pressure lift of an R32 system and more than seven times that of an ammonia system.
VOLUMETRIC CAPACITY OF VARIOUS REFRIGERANTS
The important thing to understand is that low VC refrigerants tend to operate at lower pressures and require larger system components (compressors, piping, valves, etc) while high VC refrigerants will run at higher pressures and can deliver capacity with smaller components. This introduces the first challenge when retrofitting synthetic refrigerants in existing systems: swapping a higher VC refrigerant for a lower VC refrigerant, generally, will result in a considerably undersized system. Conversely, swapping from a lower to higher VC refrigerant may compromise design pressures, scorch oil, and in most cases lead to system failure. It’s important then to know the VC of your refrigerant so you don’t waste time considering alternatives that don’t stand a chance inside your system.
Without going too far back, we can better understand the available refrigerants by looking at the refrigerants they’re meant to replace. R12 is a good place to start because it contrasts well to R22 which has twice the VC and operating pressure. Since both R12 and R22 have been phased out due to their ODP (Ozone Depletion Potential) we can track the lineage of their replacement gases to where we are today.
R12 REPLACEMENTS – “LOW VC”
R134a was the first chlorine free (ODP = 0) HFC refrigerant tested comprehensively1 and since it’s properties match closely to that of R12, it served as a good replacement and has been widely used in many medium temperature and air-conditioning applications to this day. R134a has a relatively low GWP value (circa 1300-1400 in contrasts to more than 10,000 for R12) and so has dodged some of the early US regulations on HFC refrigerants. However, the use of R134a has been limited due to the expense of building large systems with low VC refrigerants and due to restricted application in low temperatures. It has however found favorable applications with large capacity centrifugal compressors.
Even though the GWP of R134a is the lowest of all non-flammable HFCs, it hasn’t proven to be low enough for permanent use. In Europe, R134a was immediately disqualified when they required the GWP of all mobile air conditioning refrigerants to fall below 150. Europe’s F-gas phase down regulations as well as the Kigali amendment are phasing down R134a along with many other HFCs for refrigeration and air-conditioning applications as well. So the future for low VC synthetic refrigerants has hinged on the use of a different family of refrigerants, known as “HFOs” or hydrofluoroolefins.
Perhaps the most well-known and widely used HFO is R1234yf which has similar properties to R134a but with a GWP of 4. HFO’s achieve a very low GWP by being relatively unstable and breaking down quickly in the atmosphere. Long-term stability within a refrigeration system has also been a concern; however, this issue was apparently resolved through testing prior to its induction into wide-scale mobile air-conditioning (MAC) in Europe. R1234yf might have been a slamdunk R134a replacement if it weren’t for its most annoying characteristic- -flammability. Although it is being used in MAC, lingering safety concerns and controversial experiences of R1234yfinduced crash fires (Daimler-Mercedes)2 have led to further development and use of CO2 systems for MAC.
One way to get around the flammability issue with HFOs is to mix in some R134a. With enough concentration of fluorine (complements of the R134a) the flammability can be reduced to a level where “non-flammable” status can again be achieved. However, this of course comes at the cost of increased GWP and so can be viewed as a self-defeating tactic, and certainly can’t be a permanent solution. R513A has therefore been established as yet another stepping stone. Of course, as R134a undergoes a future phase down, refrigerant blends that rely on R134a will also be affected. In other words, if the Montreal Protocol is regulating R134a, by association, R513A is also regulated by it.
In time, HFOs will find their natural boundaries, as did R134a due to cost and performance concerns. For example, it’s not likely that HFOs will find wide use in cold storage or blast freezing applications as a result of their relatively high boiling points. Colder evaporating temperatures will cause systems to operate in a vacuum–drawing air and moisture into the system when there are leaks. Unlike with ammonia, even small amounts of moisture in HFO or HFC systems will degrade oil, form acids, contribute to motor burnout on semihermetic compressors, and likely freeze up expansion valves. Refrigerant price is another deterrent. The process of HFO manufacturing requires complex chemical reactions which are inherently expensive leading to prices around $35/lb in bulk, and $135/lb in smaller quantities.3 Ironically or not, a limited supply of reclaimed R12 is still available in the US to service existing systems, and it is approximately the same price as R1234yf. This suggests that HFO use in large systems may also be limited.
It is still early days for HFO refrigerants. Beyond ozone depletion and global warming, there are certain environmental issues yet to be fully understood. HFO refrigerants break down and oxidate quickly into the atmosphere as previously mentioned, however, when they do so, they turn into trifluoroacetyl fluoride and formaldehyde, but then settle out as carbon dioxide, hydrofluoric acid, and trifluoroacetic acid–also known as “TFA”4 . Researchers surprisingly don’t seem to care so much about the hydrofluoric acid as it eventually neutralizes, but TFA is known as a “durable” acid which contaminates bodies of water as it returns to the ground via rainfall. The increase of TFA in fresh water sources as a result of wider HFO use poses a real concern that has not been entirely resolved. Just like there are natural sources of CO2 emissions, there are also natural sources of TFA emissions; however, there is a limit to TFA concentration in drinking water, and some countries are already paying close attention to the additional contribution from HFOs.
In review, we’ve now looked at the implications of moving from R12 to R1234yf while making pit stops at R134a and R513A, picking up flammability along the way, and ending with a refrigerant that may contaminate drinking water and costs as much as the limited remaining supply of reclaimed R12. On the bright side, R1234yf eliminates ODP while minimizing GWP and is not expected to be more than 5% less efficient than R134a.
R22 REPLACEMENTS – “HIGH VC”
Shifting now to R22 replacements in the medium to high VC category, we can again follow the development of replacement refrigerants—which introduces some additional complication along the way. As it turns out, R22 is a good refrigerant, which in many ways behaves more like propane and ammonia than it does like much of its synthetic kind. R22 is also a pure refrigerant like propane and ammonia in contrast to its replacements, which are all blended mixtures. The early batches of HFC blends like R507a and R404A developed to replace R22 used R143a primarily mixed with R125 which were chlorine-free refrigerants and so had an ODP of 0. R143a and R125 also have similar boiling points and so these early blends were able to avoid significant temperature glide . However, with the high GWP value of R143a (4800) these early blends were regrettably not developed with GHG in mind— resulting in a consequential increase in refrigerant GWP in the first major shift away from R22.
The use of R404A and R507A has already been abandoned in Europe due to the EU’s F-gas regulations; and even in the US, these refrigerants were federally banned from use in new supermarkets for several years due to their high GWP. Further state level activity in the US is also prioritizing these refrigerants for delisting and phase down in many mainstream applications. In foresight of all this activity, a second iteration of R22 replacement HFCs were developed. While the first iteration managed to avoid substantial temperature glide, the second iteration has certainly not. Of course, to reduce GWP, the R143a had to be replaced with something else, and R32 was the logical candidate with a GWP of only 677. But refrigerant blending is never that easy; and there always seems to be a tradeoff. R32 is a flammable refrigerant (hence why it had not been used as a stand-alone refrigerant) but in weak enough concentration, the blended gas remains nonflammable. However, R32 also has a very low boiling point (-62F) as compared to R134a (-15F) which results in a large temperature glide for the final blend. R407F is one example from the 407-series refrigerant blends used as an R22 replacement, which experiences an evaporating temperature increase of more than 10 degrees (F) and a condensing temperature reduction of almost as much.
TEMPERATURE GLIDE
The debate around temperature glide has intensified since the R32 blends first came to market, and this is not surprising because of the multifaceted complications that arise because of it. Heat exchanger design and performance, system application, commissioning, and service practices are some examples of issues requiring consideration. Another is fractionation–which refers to the separation of the various refrigerants in the mixture during evaporation, condensation, or whenever liquid and vapor coexist at a constant temperature- -like in a refrigerant cylinder. This means cylinders must be charged in full to maintain refrigerant composition and performance within systems. This also means that refrigerant leaks become especially concerning if they occur in evaporators or condensers since the overall refrigerant composition can become skewed and lead to unpredictable system operation, potential degradation in capacity and efficiency, and perhaps require a full evacuation and re-charging of the refrigerant to restore performance. While this result of fractionation is a valid concern, actual experiences with evaporator and condenser leaks have been mixed and many users continue to top off refrigerant charges without validating the remaining refrigerant composition. This of course doesn’t mean that composition isn’t affected since slow efficiency losses may be hard to detect and capacity losses may not cause problems except on the hottest day of the year. Another issue with fractionation is incompatibility with certain applications. Flooded evaporator systems for example are often desirable as they offer efficiency advantages over dry expansion systems while often offering higher reliability and simpler controls. However, flooded systems are problematic for high-glide refrigerants as they would yield a different refrigerant composition in the evaporator as opposed to the rest of the system, and neither composition would match the actual desired blend.
Temperature glide also complicates system design and energy comparisons. AHRI (Air-Conditioning, Heating, & Refrigeration Institute) standard 540 requires that compressors rated for use with zeotropes (refrigerants with glide) must reference saturated dew point temperatures when representing condensing and evaporating pressures. Compressor selections are therefore often based on dew point–which is the warmest temperature during phase change. However, if dew point temperatures are also used to select heat exchangers, this will lead to oversized evaporators and undersized condensers when UA values are used which were based on the use of pure refrigerants. This is actually a major problem as there is a lack of heat exchanger rating information from manufacturers accounting for the effects of temperature glide. In fact, the common performancerating standard for evaporator air coolers (AHRI 420) excludes refrigerants with a temperature glide exceeding 2F. A large retailer in the US undersized dozens of condensers when they initially switched to R407F because they referenced compressor condensing temperatures (at the dew point) and used the UA metric they had always used which did not account for temperature glide. Because the glide of R407F is so large, several sites received condensers which were approximately 40% too small.
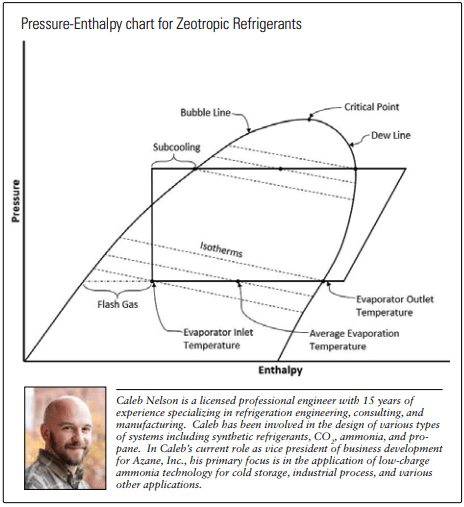
Dealing with temperature glide in evaporators is even trickier than in condensers. Averaging the bubble and dew temperatures at the evaporating pressure will unfortunately not give the average evaporating temperature. Because flash gas is produced during expansion, some of the temperature glide gets “used up” at the expansion valve, so the refrigerant entering the evaporator will actually be warmer than the bubble point temperature. Moreover, as flash gas percentage is also dependent on the amount of subcooling up stream of the expansion device, estimating your average evaporating temperature requires multiple calculation steps. This is not an insurmountable problem for designers, but it certainly complicates things for the commissioning and service technicians who often move from different refrigerants, with and without glide, in a single working day. Requiring software to accomplish what they are used to doing with a pocket PT chart while working on a roof in the rain makes this complication more than an annoyance.
PRESSURE-ENTHALPY CHART FOR ZEOTROPIC REFRIGERANTS
One critical disadvantage of temperature glide is the COP penalty it inflicts by demanding lower suction pressures. This is already well known to those who appreciate the difference between flooded systems and dry expansion systems–the latter of which require larger evaporator approach temperatures in order to generate superheat at the evaporator outlet for expansion valve control. This penalty is exacerbated by glide since even more temperature approach is needed to allow for the gliding temperature increase in addition to superheat. Some may attempt to minimize this loss by controlling to fewer degrees of superheat, but this increases the risk of liquid returning to the compressor.
Chillers operating with high flow rates and low fluid TD’s (such as ice rinks) are especially vulnerable to efficiency penalties with high temperature glide refrigerants. This is because fluid temperatures do not return to the evaporator warm enough to promote full evaporation or drive sufficient superheat at normal evaporator approaches. In retrofit cases where this has not been accounted for, systems have suffered from what’s now known as “low-TD syndrome” where an expansion valve does not sense sufficient superheat and incorrectly responds by limiting refrigerant feed to the evaporator. This reaction by the expansion valve would normally be appropriate since a loss of superheat would typically indicate excessive refrigerant feed. When superheat does not increase as a result of reducing feed, the expansion valve control continues to limit the evaporator feed until the system effectively shuts itself down. In all cases, lowering the evaporator temperature is required to fix the problem—but at the expense of system capacity and efficiency. A refrigerant with 11 degrees (F) of evaporator glide (e.g. R407F, a.k.a. Performax® LT) in a dry expansion evaporator would penalize compressor COP by 10% in comparison to a pure refrigerant operating in a flooded evaporator.
As undesirable as temperature glide may be, it appears to be a permanent fixture, at least when it comes to nonflammable synthetic (fluorinated hydrocarbon) options for replacing R22 and the other high VC refrigerants like R404A and R507A. This is because all the available “Low GWP” pure refrigerants with a higher VC are flammable and must be mixed with non-flammable low GWP refrigerants–and all of those have very different boiling points! Also interesting is that there aren’t many suitable ingredients available for mixing high VC blends for a low GWP. Primarily, there’s R32, R125, R1234yf, R1234ze(E ), R152a, and R134a, and in the correct percentages, the furthest GWP can be suppressed while remaining non-flammable is approximately 1300–which is clearly not a long-term solution. In fact, there are no low GWP (<500) halogenated refrigerants with a VC similar to R22—blended or pure—flammable or not. Refrigerant manufacturers will likely continue to look for creative ways to come up with new low-GWP options, such as mixing CO2 or propane with synthetic blends; however, doing so produces prohibitive levels of temperature glide (in excess of 20 degrees F) in most systems. In light of this, it can be concluded that there are no comparable replacements for R22 suitable for longterm use–except for natural refrigerants.
NATURAL REFRIGERANTS
Meanwhile, many of the barriers preventing a broader use of natural refrigerants (likely the same ones that instigated the first generation of halocarbons) have been overcome in the past decade, first in Europe and now in the US. For ammonia, safety concerns have been addressed through massive charge reduction in packaged systems, which also allows for easier and broader application. Limiting the risk of ammonia leaks to the outdoors and reducing the charge below 500 lbs. has significantly improved its acceptance–even encouraging several supermarket chains to use it. The many benefits of ammonia are still best exploited by medium-to-large industrial applications, and because of its high critical temperature, it’s an efficient solution in all climate zones. Cascade systems have allowed early applications with CO2 (on the low side at manageable pressures) while technology advanced to make high-pressure, transcritical systems feasible. Transcritical CO2 may become the system of choice in the US for retailers in cooler climates—as it has in Europe, Canada, and other parts of the world. Because of carbon dioxide’s low critical temperature, however, efficiency improvements have been gained with multiple stages of compression, ejectors, external subcooling, low pressure receiver overfeed arrangements, and specialized controls. Further energy benefits are available where there is sufficient heating demand which can be reclaimed from the refrigeration system. Heat reclaim can be especially beneficial for CO2 systems where it helps to improve performance above the critical point. Industrially, transcritical operating pressures have restricted the use of large compressors limiting it to commercial and light-industrial applications. Although development of larger compressors is ongoing, it is likely that ammonia (or where strategic, ammonia-CO2 cascade) will retain much of its market share as the incumbent and more efficient solution for large industrial applications.
The future of hydrocarbons is also looking brighter as the charge restrictions due to flammability concerns are relaxing. In May this year, the IEC (International Electrotechnical Commission) approved an increase from 150 grams of refrigerant per system to 500 grams. This is an indication that a similar increase in the US is now likely inevitable. Even at 150 grams, hydrocarbons such as propane have seen good market growth in smaller self-contained applications as it offers comparable performance to R22. An allowance for larger charges will no doubt be a catalyst for broader use, and perhaps could support retail applications where warm climates may prohibit the use of CO2 .
CONCLUSION
If you’re still using R22, you’re hopefully aware that its production and importation to the US ceases in a few months. However, understanding how Europe and certain industries in the US have transitioned away from R22 is helpful, especially if their hard-learned lessons can be avoided. Attempting to eliminate R22 leaks and betting on the limited supply of reclaimed refrigerant is not a long-term winning strategy— especially if everyone were to attempt it. However, is it productive to employ HFCs like R507A or R404A if it means eliminating OPD by doubling GWP? Switching to a lower GWP HFC at the expense of temperature glide and efficiency may buy time, but adds complication and running costs. At the end of the day, HFC regulation is an inevitable global trend which is gaining momentum every day—despite political pendulums. This makes it hard to ignore that there are favorable natural refrigerant options covering the spectrum from small soda machines to large industrial warehouses–especially since “freon” looks less and less favorable as it is forced to look less like R22.
REFERENCES
- Refrigerant Report 20, Bitzer Kuhlmaschinenbau GmbH, Sindelfingen, Germany, September, 2018
- IHS Markit, 2019, accessed 5-17-2019, <https://ihsmarkit.com/country-industryforecasting.html?ID=10659106922>
- Andersen, S., Nolan, M., Seidel, S., Sherry, D., 2017, HFO-1234yf: An Examination of Projected Long-Term Costs of Production, Library of Center for Climate and Energy Solutions.
- Study on Environmental and Health Effects of HFO Refrigerants, Publication number: M-917, 2017, Prepared for the Norwegian Environmental Agency, December, 2017