Avoiding Brittle Fracture When Repressurizing Carbon Steel CO2 Vessels That Have Lost Pressure
Martin Timm, Corporate Process Safety Manager and Jason Haley, CO2 Process
ABSTRACT
A vessel containing liquid CO2 may form dry ice and be at a temperature below -100°F (-73.3°C) if a leak or control malfunction causes its pressure to drop to atmospheric pressure. If the upset condition is corrected and the vessel is repressurized too quickly with warm high-pressure CO2 , there is a possibility of brittle fracture if the vessel material of construction is carbon steel. The industrial gas industry has developed safe and acceptable methods of repressurizing carbon steel CO2 vessels that have lost pressure. These are described in the Compressed Gas Association guideline CGA-6.7. This paper describes the metallurgical and thermodynamic issues, and the methods used for repressurization in the industrial gas industry.
INTRODUCTION
Carbon dioxide (CO2 ) as a refrigerant is increasing in popularity. It has many admirable characteristics, including low toxicity, non-flammability, high vapor density requiring less compressor volumetric capacity, ability to be used as a secondary coolant, ability to be integrated into an ammonia-CO2 cascade system, etc. Less desirable characteristics include:- High operating pressure
- Liquid CO2 cannot exist at pressures below 60.4 PSIG (4.2 barg) (saturation temperature -69.9°F (-56.6°C )
- The equilibrium temperature of dry ice at atmospheric pressure is very low at -109.3°F (-78.5°C)
A temperature of -109°F (-78.5°C) is well below the minimum allowable temperature for most carbon steels that are used in pipes and vessels in industrial refrigeration.
At this low temperature, carbon steel becomes brittle, and significant derating of allowable pressure is required. While at this temperature, the material is subject to brittle fracture resulting in loss of containment, especially if stressed by impact, or by shrinkage of inflexible piping arrangements.
The industrial gas industry handles large quantities of carbon dioxide in production facilities, distribution chains, and in tanks located at customer facilities. The trade associations serving the industrial gas industry have developed harmonized guidelines for safe repressurization of CO2 tanks that have lost pressure1-3. Following these guidelines allows for safe and effective restoration of the carbon dioxide vessel to normal operating conditions.
Auto-refrigeration: Ammonia vs. CO2
Liquid refrigerant in a partially full pressure vessel or heat-exchanger is usually considered to be saturated. Even if the liquid enters the vessel in a subcooled condition, with time the liquid equilibrates with the vapor space and reaches saturation conditions, at least at the liquid surface.
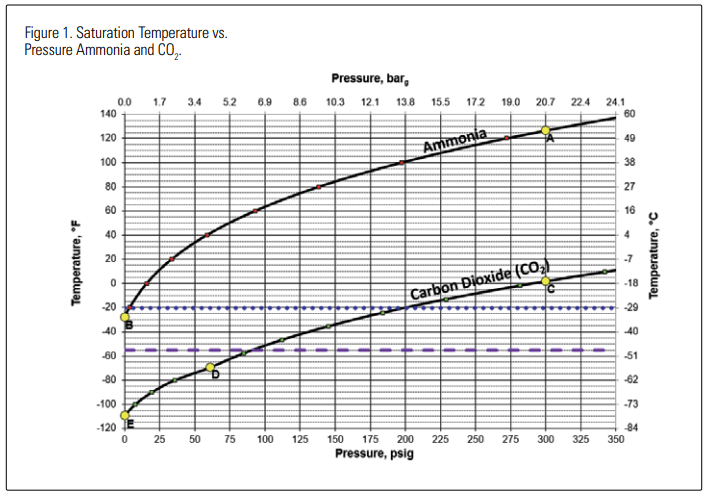
When the pressure is reduced in such a vessel, some of the liquid flashes to vapor, auto-refrigerating the remaining liquid. As the pressure drops, the pool temperature follows the saturation curve for that refrigerant. If the refrigerant is ammonia at a pressure of 300 PSIG (20.7 barg), the starting temperature is 126.5°F (52.5°C), with a lower temperature of -28°F (-33.3°C) as the refrigerant reaches atmospheric pressure. This process is depicted as moving from point A to B in Figure 1. If the refrigerant is CO2 at a pressure 300 PSIG (20.7 barg), the starting temperature is 1.7°F (-16.8°C). As the pressure of the CO2 drops, some of it evaporates, leaving cooler liquid behind, but only until the pressure reaches 60.4 PSIG (4.2 barg) at -69.9°F (-56.6°C). This process is depicted as moving from point C to D in Figure 1. At point D, the “triple point” temperature, vapor, liquid, and solid CO2 can coexist. But if the pressure is dropped even lower, all the liquid converts to a mixture of dry ice (solidified CO2 ) and vapor at a temperature below -69.9°F (-56.6°C). As the pressure of the dry ice/ vapor mixture is dropped from the triple point down to atmospheric pressure, temperature of the solid dry ice and the vapor in contact with it reaches -109.3°F (-78.5°C), depicted as moving from point D to E in Figure 1.
Minimum Design Metal Temperature (MDMT)
The horizontal dotted and dashed lines in Figure 1 represent the temperature range of -20°F to -55°F (-28.9°C to -48.3°C). Carbon steel pressure vessels normally show a minimum design metal temperature (MDMT) on the ASME nameplate somewhere in this range and are often dual stamped. The lowest MDMT shown is often no lower than -55°F (-28.9°C) or so.
The problem regarding repressurization of CO2 vessels that have lost pressure is that when the repressurization process is started, the vessel and its contents may be at a temperature below the lowest listed MDMT, and care must be exercised to avoid adding heat and raising pressure in the vapor space faster than the heat can be transferred to the cold dry ice at the bottom of the vessel. If the pressure rise occurs slowly and/or in controlled steps, the dry ice has time to absorb heat and reliquefy, equilibrating with the vapor space, and the metal walls can absorb heat and rise to a temperature where they are no longer in a brittle condition.
If the pressure rise occurs too quickly, the stratified tank may have a uniform pressure throughout, but warmer vapor at the top and much colder dry ice and/ or liquid at the bottom, in contact with the carbon steel lower shell. Thus, the vessel may be in a vulnerable condition if repressurized too quickly. “Too quickly” is a relative term. It is meant to convey the idea that the rate of change is such that contents of the vessel are able to equilibrate by heat transfer from warmest areas into the coldest areas. “Safe” rates of change are addressed in the referenced documents mentioned earlier1-3, but the preferred time scale for repressurization to the normal operating state is on the order of hours rather than minutes.
CARBON STEEL BRITTLE FRACTURE
Treatment of Embrittlement in the IIAR CO2 Handbook
The possibility of low temperature embrittlement is mentioned in Chapter 1 of the IIAR CO2 Handbook4 and also in section 16 of the sample Safety Data Sheet in Chapter 4. Chapter 7 in the handbook, the evaporator chapter, elaborates that “carbon steel is known to become brittle at temperatures below about -20°F (-28.9°C) and prone to fracture when subjected to impact loading even though the strength of the metal increases as the temperature is reduced.” Chapter 15 in the handbook, the pressure and leak testing chapter, states that “A repaired or modified section of an existing system must be above the ductile-brittle transition temperature before pressure or leak testing is carried out. Sections of pipe under insulation with residual liquid CO2 may be colder.”
Chapter 7 in the CO2 Handbook states that stainless steel “is not susceptible to embrittlement even at extremely low (cryogenic) temperatures,” and provides similar guidance for copper. So, clearly, IIAR’s CO2 Handbook has multiple statements alerting the reader to the possibility of embrittlement of carbon steel when exposed to temperatures approaching the triple point temperature and temperature of dry ice at atmospheric pressure. While the alerts are valuable and appropriate, however, they do not provide specific guidance on how to deal with a pressure vessel that has lost pressure and is in a low temperature condition.
Incident Reports
Failure of a CO2 pressure vessel is a relatively rare event, and failure due to repressurization from a cold condition is even more rare, possibly due to the rarity of a total loss of pressure and the industrial gas industry having guidelines in place on how to safely repressurize. The authors were unable to find any published reports regarding failure of a CO2 pressure vessel during repressurization. The following references are included because they provide good background information on CO2 vessel design.
In one of the earliest reports found, Copeland and Wishart5 from Sandia Laboratory discussed issues around coarse-grained carbon steels and speculated on metallurgical aspects related to the reported failure in 1974 of a storage vessel at a CO2 production facility. They stated that original press reports suggested the initial failure was of an ammonia vessel, but they concluded the vessel that failed first probably contained CO2 . Copeland and Wishart reported that Sandia evaluated the several CO2 tanks they had in service at the time and, based on considerations of potential brittle fracture, elected to replace a vessel made from coarsegrained A515 steel with a new tank manufactured from A516 steel. None of the vessels discussed appear to have been used in a closed-cycle CO2 refrigeration system.
Zhang and Schork6 describe four incidents in which CO2 vessels catastrophically failed, including two in which brittle fracture played a role, but those scenarios involved overfilling or overheating of trapped contents rather than intentional repressurization from a cold, low-pressure state. None of the four incidents involved vessels used in closed-cycle refrigeration systems. One of the four vessel failures is described in detail by Clayton and Griffin7.
Evaluation Methods
Engineering literature in various industries address concerns about brittle fracture of carbon steel vessels and piping systems. The following references are representative of publications that examine the analytical methods that can be applied to designing and operating vessels within a safe range. Such considerations are the foundation for the CO2 vessel repressurization methods commonly used in the industrial gas industry.
Riley provides a general discussion of stresses in ammonia refrigeration piping and of the requirement for flexibility analysis and/or the derating of certain materials in piping systems operating below -20°F (-28.9°C).
Kumar9 provides a detailed discussion of brittle fracture metallurgy and selection of materials for hydrocarbon applications when low-temperature excursions are infrequent but possible. He includes references to applicable ASME and API guidance documents.
and API guidance documents. King
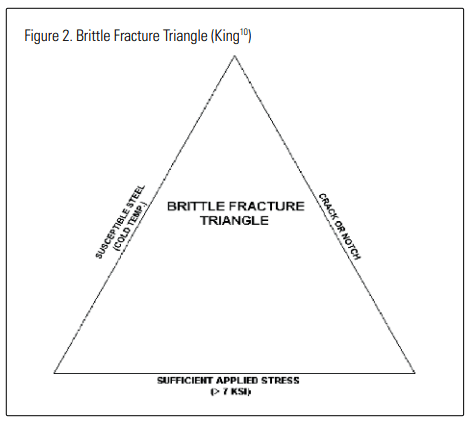
states that the following three conditions are necessary for brittle fracture to occur:
- A susceptible steel (susceptibility increases as temperature decreases);
- A stress riser, such as a crack or a notch (as often is found in weld defects);
- A sufficient applied stress above a minimum stress level (~7000 psi [48MPa] for carbon steels).
He represents this graphically with the diagram reproduced in Figure 2. King goes on to recommend an analysis approach and provide examples. His approach is consistent with the methods embodied in the discussion and recommendations in CGA Guideline G-6.7 and by AIGA and EIGA in their harmonized documents.
Pragar provides a detailed review of the history of impact testing exemption curves, and makes a strong argument that the curves in the ASME BPVC Section VIII are too conservative for steels made with modern manufacturing methods.
Banac et all discuss brittle fractures in ammonia-related industries. They discuss API methods for vessel evaluation regarding brittle fracture potential and describe some incidents that have occurred in the ammonia production industry. Like King, Banac et al discuss the three conditions necessary for brittle fracture, but they also state that high residual stresses can be present due to welding or heat treatment, even before the application of internal pressure.
CO2 TEMPERATURE AND PRESSURE VS. TYPICAL CARBON STEEL VESSEL RATING
A useful technique when discussing repressurization methods is to superimpose allowable temperature vs pressure for a given vessel on a graph like that shown in Figure 1. Figure 3 shows such a graph for a “sample” CO2 storage vessel constructed from normalized A516 carbon steel, a common material of construction for carbon dioxide storage vessels, and capable of holding 30 tons of CO2 . The boundary for the brittle zone was calculated as recommended in ASME BPV Code13, including section UCS 66. Note that this graph is specific to the material, and wall thickness assumed for this example is representative of the trends that are observed. Similar graphs can be constructed for vessels manufactured with different materials, wall thicknesses, heat treatments, etc., and many combinations will result in making the vessel either more or less susceptible to brittle fracture.
For this sample vessel, a dramatic reduction in allowable pressure is seen at temperatures below about -50°F (-45.5°C). Below -50°F (-45.5°C), the carbon steel is in a brittle condition. It is desirable to avoid operating in the cross-hatched area (below the solid black line) due to the possibly of brittle fracture of the material.
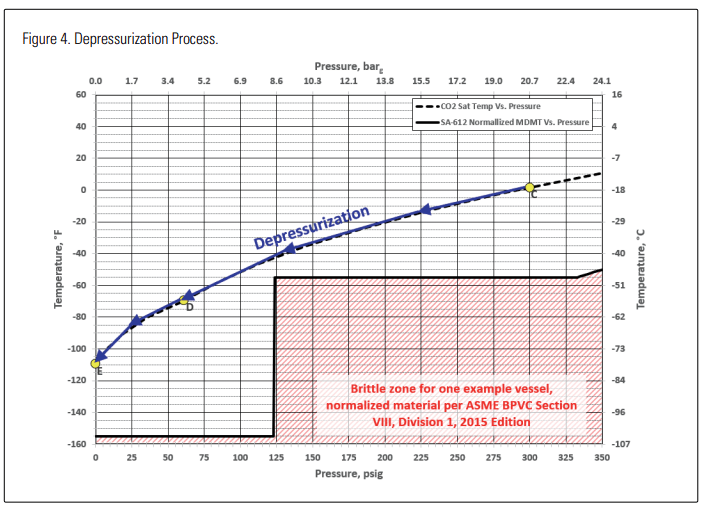
Now consider what happens if the contents of such a vessel are depressurized. Figure 4 illustrates the reduction in temperature as the vessel pressure drops. In this case, for this A516 vessel, the saturation temperature of the CO2 at a given pressure never drops below the vessel’s minimum allowable temperature for that pressure. There is little or no danger of brittle fracture as the vessel is depressurizing.
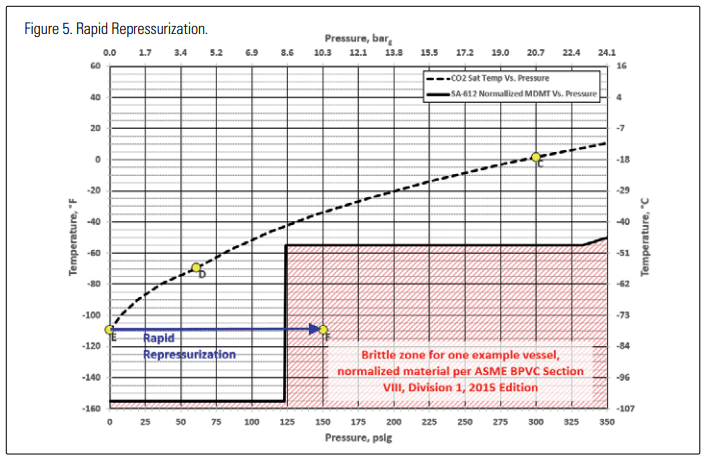
Let’s now consider a couple of pathways that could occur during the repressurization process. Figure 5 shows a rapid repressurization process. It assumes that a source of relatively warm carbon dioxide gas is added to the head space at a rapid rate, causing the pressure to rise quickly, creating a non-equilibrium condition within the vessel.
Given enough time, the warm gas would equilibrate with the cold dry ice and/or cold CO2 liquid in the vessel, but initially, near-equilibrium conditions only exist near the surface of where the gas is contacting the liquid pool or residual dry ice. If the repressurization has raised the pressure in the head space to 150 psig (10.3 barg), but there is residual dry ice in the bottom of the vessel, instrument lines, etc., that dry ice may still be at -109°F (-78.3°C), as illustrated by point “F” in Figure 5. From this figure, it can be seen that point F is in the “brittle region,” so those portions of the vessel at that combination of pressure and temperature may be vulnerable to brittle fracture.
Raising pressure at a “rapid rate” may be possible in a large industrial refrigeration system where multiple evaporators are operating in parallel and high pressure defrost gas is available from a central machinery room. If this gas is introduced in an uncontrolled fashion to a small “iced” vessel in the system, it might be possible to increase the pressure in the vessel very quickly. On the other hand, in small or simple systems that don’t utilize multiple evaporators, it may be difficult to generate enough high-pressure gas to effect repressurization at even the desired slower rate, much less a fast rate. In general, the desired time scale of repressurization to avoid the brittle fracture range is on the order of hours, not minutes.
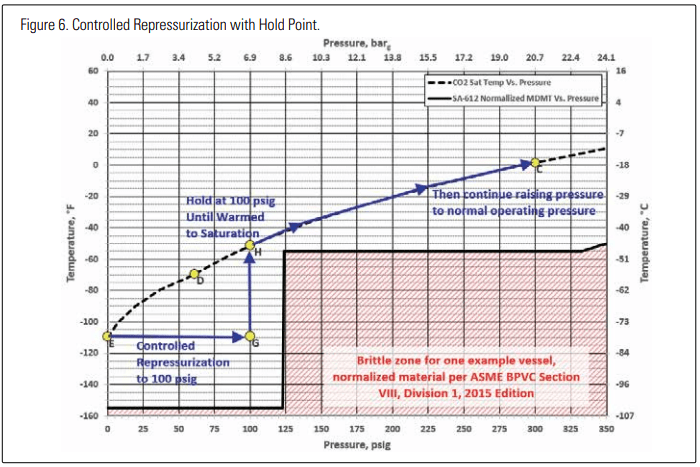
Next, let’s consider an alternate repressurization process, as shown in Figure 6. In this process, warm gas is added to the head space of the vessel to raise its pressure to 100 PSIG (6.9 barg) (point G), and then the pressure is held there for enough time to reach nearequilibrium between the head space and any liquid or dry ice in the bottom of the vessel, eventually reaching point H. Further addition of warm gas or heat to a vessel containing only liquid and vapor would then be expected to follow the saturation curve upwards towards point C. The recommended methods in CGA 6.7 and harmonized industrial gas industry documents are similar to the one shown in Figure 6.
The recommended methods include variations on how heat is added back into the vessel in the form of vapor or liquid. Further details are provided below.
WAYS A CO2 VESSEL CAN ICE IN A CLOSED CYCLE REFRIGERATION SYSTEM
Carbon dioxide has the unique physical property of forming a solid phase in equilibrium with a vapor phase, with an absence of liquid phase below a certain pressure. This condition occurs when the pressure of liquid carbon dioxide is allowed, either intentionally or accidently, to be reduced below its triple point pressure of 60.4 psig (4.2 barg). When this occurs, the liquid contents within a tank or vessel are converted to a mixture of solid carbon dioxide and vapor. When this happens in an undesired condition it is commonly referred to as “icing” the tank or vessel.
Leaks
One cause of liquid content losing pressure and subsequent “icing” of a tank is loss of pressure due to a leak to the atmosphere. Gasket failures due to improper selection and/or installation, cracks in piping or vessels, pressure relief valves “sticking open,” and/or leaks at threaded connection are common causes of icing. Valves accidently left open during maintenance, or opened incorrectly during operation, also can cause a loss of pressure leading to formation of dry ice.
Control Failures
Within a CO2 refrigeration system, the pressure of a CO2 refrigerated evaporator may be managed by a control loop incorporating a downstream CO2 compressor. Failure of this control loop could result in lower than desired operating pressure. If this event were to continue, low pressure cut-out devices should de-energize compression equipment prior to it reaching an unsafe operating pressure. However, if these systems were to fail and evaporator pressure was allowed to drop below 60.4 psig (4.2 barg), the result would be formation of dry ice in the evaporator.
Charging Liquid CO2 Directly into an Evacuated System
During initial charging of a CO2 refrigeration circuit, pressure should be raised to above the pressure corresponding to the MDMT of the vessel using gaseous CO2 . If liquid CO2 is “sprayed” into piping or vessels that are at a pressure below the triple point pressure of 60.4 psig (4.2 barg), the liquid will flash into dry ice “snow” and vapor. As the dry ice snow deposits within the vessel, surfaces may be cooled down below the MDMT and stresses may be created due to differential thermal contraction of the metal.
REPRESSURIZATION OPTIONS IN G-6.7
The industrial gas industry, as outlined in CGA G-6.7 “Safe Handling of Liquid Carbon Dioxide Containers That Have Lost Pressure” has recommended four methods as safe practices to repressurize a container in an upset condition. These methods are based on decades of experience using carbon steel vessels in liquid carbon dioxide storage applications. The following descriptions are only summaries. Reader are highly encouraged to obtain CGA G-6.7 if in the USA, or the harmonized versions in regions outside the USA.
Method 1 – Unassisted Natural Repressurization
In this method, the container is isolated and pressure within is allowed to rise to no more than 100 psig (6.9 barg) until all signs of dry ice are gone. During this time, ambient heat slowly warms the contained CO2 causing the dry ice to melt and resulting liquid to warm to normal operating temperatures.
This method has the disadvantage of being extremely slow depending on the inventory of solidified CO2 , ambient temperature, and quality of the insulation on the container. This method does, however, offer the benefit of being a selfregulating process.
Method 2 – Hot Gas Warming at No Pressure
This method begins by removing the container from service and any venting and remaining pressure in the container. Pressure should be removed through a process of first draining any residual liquid carbon dioxide from the tank through a liquid connection. After all liquid carbon dioxide is removed from the container it should be depressurized completely through a vapor connection. With the tank now under no pressure, large quantities of warm, dry air or gas may be introduced to the container through an open manway or connection and vented to atmosphere. The heat within this gas will cause the dry ice to sublime to vapor carbon dioxide, which will then be carried away along with the purge gas.
The major advantage of this method is that there is no risk of container rupture, as all warming is conducted with no pressure on the container. As far as disadvantages, the hot gas warming method can cause large volumes of carbon dioxide to be released as the dry ice is sublimed. If not vented correctly this can create an asphyxiation hazard. Also, prior to returning the container to service, it must be cleaned and purged to remove moisture and contaminants that were introduced via åthe purge gas. Finally, this method may necessitate entry through a manway, which would require confined space entry restrictions and procedures.
Method 3 – Carbon Dioxide Gas Pressurization up to 100 psig (6.9 barg)
This method is commonly the first step of a two-part process, in which this approach is subsequently followed by Method 4, outlined below.
This method attempts to repressurize the container to 100 psig (6.9 barg) using an external source of vapor carbon dioxide and therefore partially liquefy the dry ice held within the depressurized container. The external source of carbon dioxide, bulk liquid cargo trailers or other, is connected to the depressurized container and allowed to send vapor carbon dioxide. This vapor will condense against the dry ice, simultaneously causing the dry ice to melt. The resulting liquid level in the tank will rise to cover the dry ice at which time the incoming vapor will rapidly pressurize the tank.
Method 3 offers a more rapid way to repressurize the container to 100 psig (6.9 barg) while maintaining the container below its minimum stress level for brittle fracture.
All of this is accomplished without the loss of carbon dioxide from the system. This method does require further action, as on its own it does not provide enough warming to the tank to ensure all dry ice held within the container has been melted. Further warming and recirculation of the liquid per Method 4 is recommended.
One disadvantage of this method is that if a system is already at the proper charge level when the icing event happens, it will be overcharged with CO2 once the thaw is completed unless the source of carbon dioxide gas is from within another part of the system. CO2 evaporators already equipped with hot gas defrost are ideally suited for this method, but if constructed from carbon steel the pressure should be limited to 100 psig (6.9 barg) until all dry ice has been liquified and temperatures raised back above the MDMT.
Method 4 – Recirculation of Warmed Liquid
This method should be preceded by Method 3 unless the container is already at or above 100 psig (6.9 barg). In this method liquid carbon dioxide is pumped from the tank, sent through an external heater, and then returned to the tank, causing any remaining dry ice to melt and promoting a more uniform temperature throughout the tank contents. This circulation operation can be stopped after the container pressure has reached a pressure exceeding the pressure coincident with the MDMT of the container. If the pressure condition remains stable, indicating its contents are at or near equilibrium, the container may be returned to service.
Method 4 offers a relatively rapid method for returning a container to normal service while minimizing the risk that the vapor pressure in the container will exceed the equilibrium pressure of the circulating liquid. During this procedure, no carbon dioxide is released from the system. This method does require sourcing, setting up, and energizing additional equipment with minimal connection requirements. The additional equipment may or may not be readily available at the typical industrial refrigeration facility.
The four methods summarized were originally recommended for liquid carbon- dioxide storage applications and based on experiences in that industry segment. However, the advantages and disadvantages of each are still relevant when evaluating for use in a closedcycle CO2 refrigeration application. The closed-cycle CO2 refrigeration application also brings with it some additional or different criteria.
Comments on the Four Methods
One important criterion is the acceptable time until the equipment is brought back online. If part of a multi-unit refrigeration system in which running without a single unit is acceptable, Method 1 may provide a simplistic and self-regulating option, without the need of external equipment, gas sources, or outside resources.
Another aspect that must be considered is the complexity of the equipment that has depressurized. In a closed-cycle refrigeration system, the equipment that has iced may not just be a simple vessel. It may consist of heat exchangers, receivers, etc., all needing to be safely returned to pressure. With this type of equipment comes additional challenges such as restrictive geometries posed by tubes and baffles in heat exchangers, or additional working fluids within the system, as is the case with ammonia/ CO2 cascade systems.
Method 3, controlled repressurization with warm gas, is probably the most widely applicable for the industrial refrigeration industry due to a familiarity and comfort level with hot gas defrost of evaporator coils. The concepts and practicalities are similar.
Each refrigeration end user should evaluate their system to determine what system limitations exist, which criteria are most important to them, and which repressurization methods best fit their system. These should then be appropriately documented in standard operating procedures and operating manuals.
Consideration of which repressurization method to be used should not be limited to end users. Planning for the possible need for repressurization of a system that has lost pressure should start back in the system design phase. The inclusion of appropriate nozzles and connections to facilitate a recommended repressurization method should be considered.
CONCLUSIONS AND RECOMMENDATIONS
The risk of “icing up” a pressure vessel or heat exchanger in CO2 refrigeration service is credible but of low probability. Many owners of CO2 refrigeration systems and their contractors will never experience this phenomenon. Still, owners and their contractors should know what to do if it happens.
Repressurization can be done safely with methods similar to industrial gas industry methods detailed in CGA G-6.7 and internationally harmonized regional versions.
The authors of this paper recommend that industrial users review their refrigeration system to determine if carbon steel vessels and heat exchangers are being used for carbon dioxide, and whether there are credible scenarios for a loss of pressure that would cause the CO2 temperature to drop below the minimum design metal temperature (MDMT). If a credible threat of pressure loss exists, the following steps, if not already taken, should be considered:
- Include these scenarios in process hazard analysis documentation (HAZOP, what-ifs, etc.).
- Include appropriate cautions and warnings in operating procedures and maintenance procedures
- Obtain CGA G-6.7 or one of the harmonized versions for your region and studying the safety information and guidelines provided.
- Assemble a task force to examine how methods best suited to storage tanks in the industrial gas industry might be modified for the needs of the industrial refrigeration industry and applied in an industrial refrigeration facility.
- Train personnel on proper steps to take if repressurization of an “iced” system must be performed.
In addition, the authors of this paper suggest that IIAR consider including recommendations for design details that might facilitate vessel and heat exchanger thawing/warming in future editions of the CO2 Handbook and related IIAR documents.
REFERENCES
- Compressed Gas Association (CGA). CGA G-6.7 Safe Handling of Liquid Carbon Dioxide Containers That Have Lost Pressure. Chantilly, VA: Compressed Gas Association; 2009.
- European Industrial Gas Association (EIGA). IGC Doc 164/10/E Safe Handling of Liquid Carbon Dioxide Containers That Have Lost Pressure. Brussels, Belgium: European Industrial Gas Association; 2010.
- Asian Industrial Gas Association (AIGA). AIGA 074/11 Safe Handling of Liquid Carbon Dioxide Containers That Have Lost Pressure. 4th ed. Singapore: Asia Industrial Gas Association; 2009.
- IIAR. CO2 Handbook. Alexandria, VA: International Institute of Ammonia Refrigeration; 2018.
- Copeland EH, Wishart R. Pressure Safety Aspects of Liquid CO2 Storage–A Case History. Albuquerque, NM.; 1976.
- Zhang Y, Schork JM, Ludwig K. Revisiting the Conditions for a CO2 Tank Explosion. 9th Glob Congr Process Saf. 2013:1-12. http://www. allriskengineering. com/library_files/ AIChe_conferences/AIChe_2013/ data/papers/Paper292075.pdf.
- Clayton WE, Griffin ML. Catastrophic failure of a liquid carbon dioxide storage vessel. Process Saf Prog. 1994;13(4):202-209. doi:10.1002/prs.680130405
- Riley T. Pipe Stress or Flexibility Analysis in Refrigeration Piping. In: IIAR Proceedings. Nashville, TN: IIAR (International Institute of Ammonia Refrigeration); 2000. www. iiar.org.
- Kumar S. Material selection for low- temperature applications. Hydrocarb Process. 2004;(July).
- King RE. Auto-refrigeration/brittle fracture analysis of existing olefins plants— translation of lessons learned to other processes. J Hazard Mater. 2007;142(3):608- 617. doi:10.1016/j.jhazmat.2006.06.066
- Prager M. Impact Testing Exemption Curves for Low Temperature Operation of Pressure Piping. In: IIAR Proceedings. Dallas, TX: IIAR (International Institute of Ammonia Refrigeration); 2009. www.iiar.org.
- Benac DJ, Shaffer D, Wood D. Managing Cold Temperature and Brittle Fracture Hazards in Ammonia-Related Industries. In: Ammonia Plant Safety Symposium. ; 2015.
- ASME. Boiler and Pressure Vessel Code. Section VIII, Division 1; 2015.